Goals of Chapter Assess heat transfer associated with changes in temperature and changes of state. Apply the First Law of Thermodynamics. Define and understand. - ppt download
Energy equivalent of 10.00 cm^ 1 is :a) 2.0×10^ 22J per photon b)28.6×10^ 3kcal mol^ 1 photon c)12.0×10^ 2 kJ mol^ 1photon d)all of the above
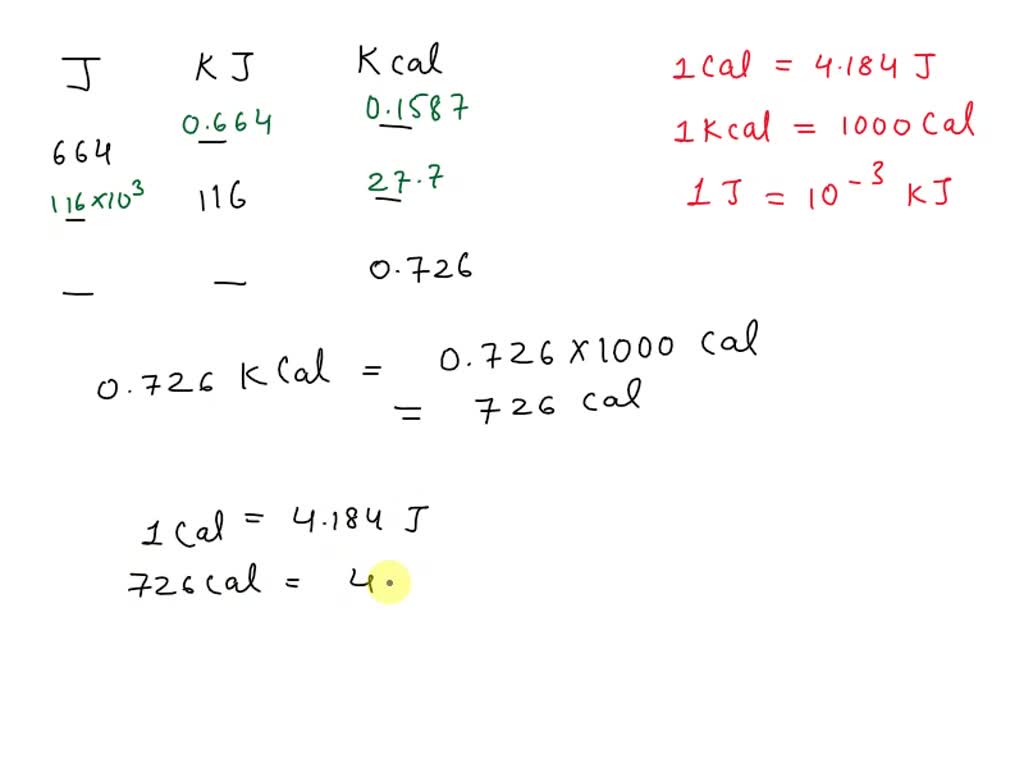
SOLVED: Expressing amounts of energy in different energy units is necessary to solve many chemistry problems. For practice, complete the following table. The Joule (J) is the SI unit of energy. 1

Chapter 6. Calorie One calorie expresses the quantity of heat necessary to raise the temperature of 1 g of water by 1° Celsius. Kilocalorie (kCal) - ppt download